Noncoding RNA in Development and Disease
Short description
Chandrasekhar Kanduri's lab is primarily interested in understanding the functional role of long noncoding RNAs (lncRNAs) in cancer development and progression. Using in-house developed functional screens, we have characterized several lncRNAs with functional implications in cancer. Using state of the art technologies we will characterize the mode of action of lncRNAs during normal and cancer conditions. We hope our future efforts will take some of these lncRNAs into clinics.
PAN-cancer approach to study functional long noncoding RNAs
Chronic cell proliferation is considered one of the important features of cancer. Cell division cycle plays a critical role in the regulation of normal and chronic cell proliferation by responding to cell cycle regulators (cyclins, cyclin dependent kinases (CDKs), growth factors, growth suppressors, tumor suppressors and oncogenes). Cell division primarily comprises two phases: the DNA synthesis (S) phase and the mitotic (M) phase. The pathways that control DNA synthesis or replication during S-phase are critical for normal cell proliferation, as errors during DNA synthesis could result in abnormal cell proliferation. Importantly, replication occurs during different stages of S-phase. For example, replication of active chromatin regions occurs during S-phase whereas that of heterochromatin or repressive chromatin regions occur during late S-phase. Thus, characterization of mechanisms that control S-phase progression would allow us understanding the role of S-phase in normal and disease conditions. In comparison to protein-coding genes, the role of lncRNAs in S-phase regulation is still poorly understood.
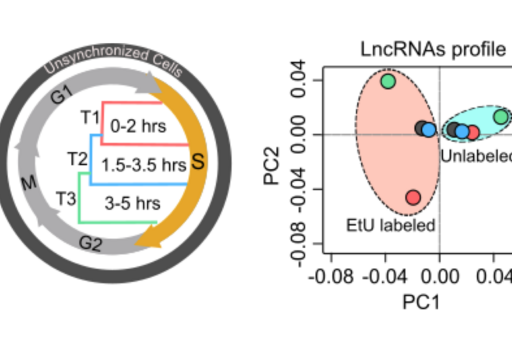
To explore the functional role of lncRNAs in S-phase progression, we need to produce time resolved transcription maps covering early, mid and late stages S-phase. To this end, we have adapted a real-time strategy "Nascent RNA Capture assay", whereby we can capture the ongoing transcription of lncRNA with high turnover in different compartments of S phase. The EtU labeled RNA along with unlabeled total RNA, representing steady state levels, collected at early, mid late stages of S phase, are subjected to deep sequencing. By applying state of the art computational tools on Etu labelled and unlabelled steady state RNA levels from early (T1), mid (T2) and late (T3) S-phase stages and unsynchronized HeLa cells, we identified 1,145 S-phase-specific temporally expressed lncRNAs (Meryet-Figuiere M, et al. Cell Cycle 2014).
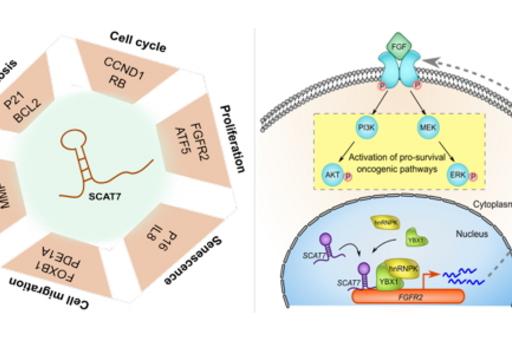
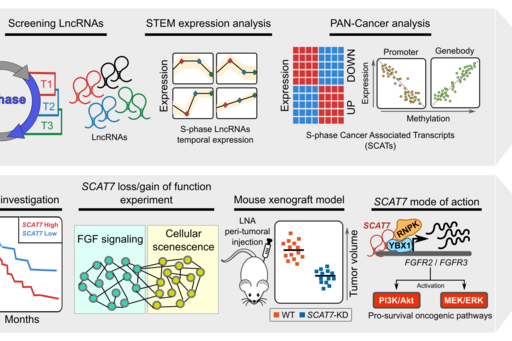
By analyzing their expression levels in 6,419 solid tumors and 701 normal tissue samples from The Cancer Genome Atlas database covering 16 cancer types, we identified 570 differentially expressed lncRNAs at least in one cancer type. Likewise, our systematic and detailed clinical investigation using 14 cancer types, identified 633 independent prognostic markers. Our detailed mechanistic investigations on one of the SCATs SCAT7 revealed that it is oncogenic lncRNA, which interferes with all well characterized cancer cell hall marks (Ali MM, et al. Nature communications 2018). Thus these S-phase-specific cancer-associated lncRNAs (SCATs) would serve as huge resource to explore their functional role in tumor initiation and development.
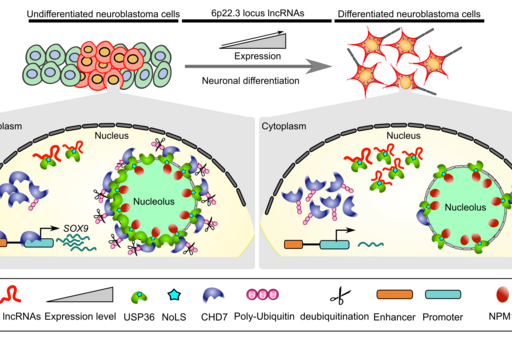
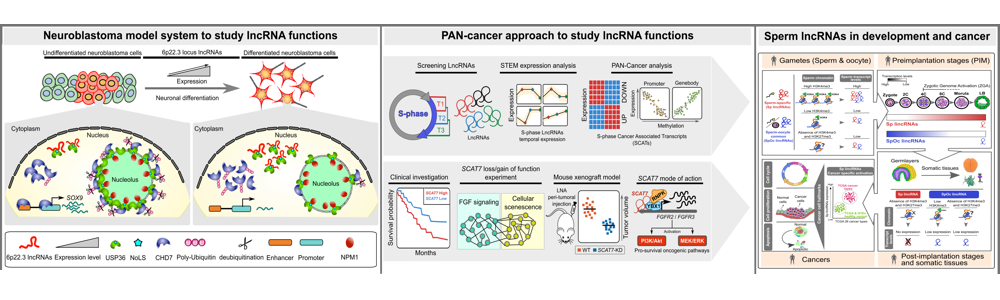
Neuroblastoma model system to study functional long noncoding RNAs
CASC15: By analyzing the RNA-sequencing data of high- and low-risk neuroblastoma tumors we identified several differentially expressed lncRNAs. Some of the top differentially expressed lncRNA genes (NBAT1, CASC15 and its isoforms CASC15-003 and CASC15-004) map to the neuroblastoma risk-associated locus 6p22.3. Several single nucleotide polymorphisms located in the 6p22.3 locus was implicated in the metastatic high-risk neuroblastoma by genome wide association studies. However, lack of any known protein coding gene in this locus made it challenging to functionally characterize the locus. We found that both NBAT1 and CASC15 (CASC15-003 and CASC15-004) are pro-neurogenic lncRNAs with high expression in Neural Crest (NC) cells and their deficiency blocks differentiation of both NC and neuroblastoma cells. Consistent with their role in neuronal differentiation lower expression of 6p22.3 lncRNAs correlates with poor prognosis of neuroblastoma patients. Based on our published evidence, NBAT1 and CASC15-003 share similar mechanisms that underlie tumor suppression. We have shown that NBAT1 and CASC15-003 interact with a deubiquitinase protein USP36 and regulate the nucleolar localization of USP36, thereby interferes with the proteasome mediated degradation of chromatin remodeler CHD7. During differentiation of neuroblastoma cells spatial distribution of USP36 is altered: nucleolar localized USP36 gets dispersed in the nucleus but in the neuroblastoma cells lacking NBAT1 and CASC15-003, USP36 remains in the nucleolus. Nucleolar USP36 promotes CHD7 stabilization which activates the expression of pro-tumorigenic genes including SOX9 (Mondal T, et al. Cancer Cell 2018).
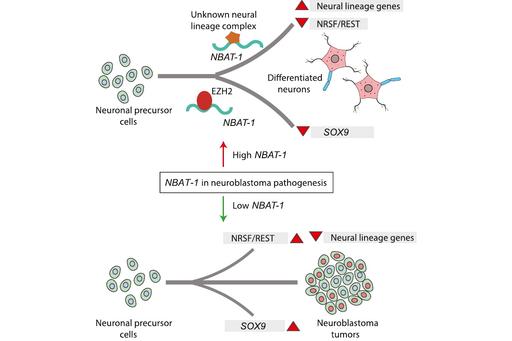
NBAT-1: is differentially expressed between low and high-risk tumors. Its lower expression correlates with poor prognosis in neuroblastoma patients. It controls neuroblastoma progression through regulating pathways implicated in cellular proliferation, invasion and neuronal differentiation. Model explaining the tumor suppressor and neuronal differentiation properties of NBAT-1. NBAT-1 functional interaction with EZH2, a member of PRC2 complex, controls tumor progression by suppressing protumor genes such as SOX9, VCAN, and OSMR via regulating chromatin structure. On the other hand, NBAT-1promotes neuronal lineage commitment by suppressing NRSF/REST by interacting with an unknown neuronal-linage-specific transcriptional repressor. The NBAT-1/EZH2 interaction has no functional role in the repression of NRSF/REST (Pandey GK, et al. Cancer Cell 2014).
Thus we are using these two lncRNAs to understand the lncRNA dependent tumor suppressor mechanisms using neuroblastoma as a model system.
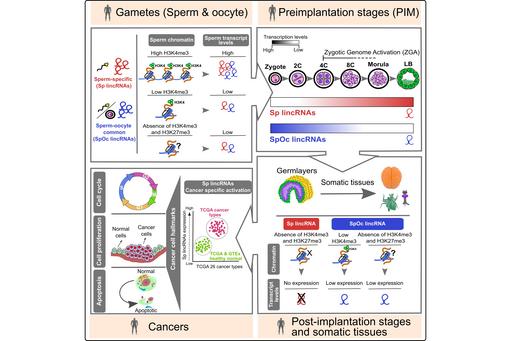
Sperm originated long noncoding RNAs in development and cancer
For long, sperm considered a passive vehicle, mere transmitting the paternal genome into oocyte during fertilization. Contrary to the prevailing view on sperm, recent studies have shown that sperm chromatin can influence gene expression during preimplantation development. However, the importance of sperm encoded transcripts and chromatin imprints in organismal development is poorly investigated. In this study, we show that human sperm and oocyte carry equal number of transcripts. Sperm and sperm-oocyte transcripts carry distinct chromatin structures, correlating with their transcript levels. Sperm-specific transcripts are not expressed in early preimplantation embryos but become activated during the onset of zygotic genome activation, whereas sperm-oocyte expressed transcripts remain active during early preimplantation development but starts to degrade at the onset of zygotic genome activation. Interestingly, sperm-specific lincRNA promoters carry distinct chromatin structures that are exclusive to sperm and not present in the embryonic germ layers and somatic tissues. More importantly, sperm-specific lincRNAs showed cancer-specific activation (Subhash S, et al. iScience 2020). Hence we are particularly interested in understanding the functional role of sperm-specific lincRNAs in cancer initiation and progression.
Chandrasekhar Kanduri
Principal Investigator
Affiliation:
Department of Medical Biochemistry and Cell Biology,
Institute of Biomedicine
Group members
Current lab members:
Silke Reischl
Mirco Amerigo Di Marco
Prasanna Kumar Juvvuna
Sagar Mahale
Subazini Thankaswamy Kosalai
Tanushree Ghosh
Bharat Prajapati
Mohamad Moustafa Ali
Santhilal Subhash
Alumni lab members:
Tanmoy Mondal
Daniel Jachimowicz
Sanhita Mitra
Kankadeb Mishra
Luisa Statello
Vijay Suresh Akhade
Somsundar Veppil Muralidharan
Sara Wernig-Zorc
Caroline Miranda
Arif Mohammad
Gaurav Kumar Pandey
Matthieu Meryet-Figuiere
Alva Rani James
Roshan Vaid
Björn Reinius
Natalia Papadopoulos
Faizaan Mohammad
Nupur Thakur
Radha Raman Pandey