- Home
- Research
- Find research
- PPI example: Patient and public involvement in a research group's steering group
PPI example: Patient and public involvement in a research group's steering group
Senior Professor and former Centre Director of GPCC Inger Ekman talks about her experience of patient and public participation (PPI) in research.
Research project: Several projects over many years, one example is: IHOPe - Integrating Health promotion with and for Older People-eHealth.
Type of project: The IHOP-e project includes a randomised controlled evaluation, a process evaluation and a health economic evaluation.
Project time: 2018-2023
Interview with: Inger Ekman, Senior Professor in health sciences, founder and former Centre Director, GPCC and P.I. for a number of projects including IHOP-e.
In which project(s) have you had PPI representatives?
I have always tried to have PPI representatives in my own studies, and have worked with them in other projects as well. In GPCC's position paper Person-centred care – Ready for Prime Time written in 2011, we described person-centred care as an ethical standpoint that guides our practical actions as fellow human beings and professionals. This was in line with the European Patient Forum’s campaign for the patient's role as a "co-producer" and partner in their care and rehabilitation.
In my current research group, we collaborate with two PPI representatives.
How many PPI representatives do you usually have?
It is good if there is more than one, then they have each other for support. There are always several scientists. It is also important that they are involved from the beginning and throughout the process. They are helpful when planning the study, for example they help to formulate relevant and clear research questions, but also in the selection of questionnaires, number of questions, information material for research participants, and so on. During the test and pilot phase, there is an extra amount of issues we need to discuss with our PPI representatives, while there are fewer questions after the pilot and during the data collection.
How did you recruit them?
It has not always been easy to recruit PPI representatives, but we have done it via patient organisations first and foremost. For example, The Swedish Heart and Lung Association has an education programme for its members which prepares them for participating in research projects and teaches them a little about, for example, different types of study methods etc. For example, we recruited PPI representative Björn Störtebecker that way, who then also became a member of the GPCC person council for patients and relatives/carers. It's good when they're a little prepared and interested.
However, not all of our PPI representatives have been recruited through patient organisations. Some can be found through networks, for example, one of the ones we have now is a next door neighbour to one of our researchers. Sometimes it can be difficult due to a patient group's circumstances, for example if they have restricted levels of energy. Participating in the research group must not become a stress factor.
In which part/s of the research cycle have they participated?
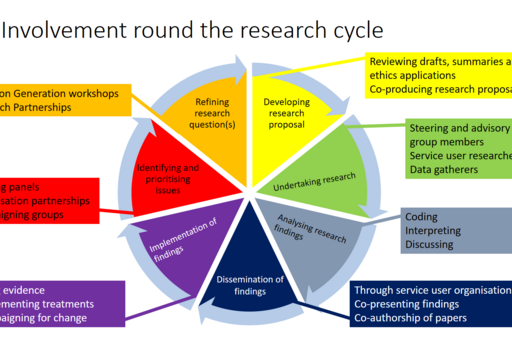
Patient involvement throughout the cycle is important, but especially when designing the study. PPI representatives need to see if the project's purpose and research questions are reasonable. Even when designing questionnaires, their input is important; is it possible to understand and answer the questions? Are there too many questions? The representatives we have included have been very helpful in this, even when it concerns validated questionnaires. It has mainly proved to be a risk that you have too many questions.
PPI representatives can also help us assess whether research issues and other issues are relevant in different types of applications for research grants. Then we usually talk about it, reading long applications can feel heavy and difficult.
PPI representatives can also make a significant contribution to information about the study. In our latest project IHOP-e Person-centered remote care for frail elderly people, our PPI representatives have been involved in reviewing letters and information material. We have received very good views on whether these are understandable and relevant.
It is not always relevant for PPI representatives to participate in every stage of a project. The same applies to researchers. If you have produced a good design and protocol, that is, the implementation plan, and especially after the pilot is completed, there are also some researchers who do not participate in our research group meetings every week, but less often. But even at this stage, problems can arise, for example, it can be difficult to recruit patients. Then we can ask the PPI representatives what they think is the reason behind it.
PPI representatives have also participated in our projects as co-authors; they have then read and commented on the script.
How do you work with your PPI representatives in practical terms?
They are invited to all our weekly meetings with researchers from different disciplines such as pharmacists, occupational therapists, doctors, nurses and research assistants. There we do different things; for example, read a book chapter or a scientific article. PPI representatives participate when they think they have time and when the agenda feels interesting and relevant to them.
Usually we have had at least one PPI representative per project. They contribute with individual experiences. For example, in the IHOP-e project, one PPI representative also has a large network of friends to whom she takes certain questions further. "I'm going to ask my crew," she says. That is great.
Then there may be periods when PPI representatives may be busy with other things, such as caring for a grandchild. Especially in such circumstances it is good that you have more than one PPI representative.
It is important that they do not feel pressured to attend all meetings. They receive compensation for travel and expenses.
Did any problems/difficulties arise?
I think the challenge is to recruit. Finding the right people. Then once you find the right people, there are no problems.
What tips would you like to give other researchers who are thinking of involving patient researchers?
It is preferable to involve them very early on. Even when you are preparing a study. You gain a lot from that.
Nor should one be afraid that they may seem too ill. For example, in one study we had a workshop where we stayed in a hotel overnight and we had PPI representatives with us. I especially remember one person. We were worried that this person might be too ill, but she knew herself what she could cope with and she contributed a lot. We also had a great time together.
Is there anything else you want to bring up?
It is important to have mutual respect. You also have to be clear with each other, and have clear meeting agendas. PPI representatives in our project team are invited to all meetings, but learn to decide for themselves what interests them.