Analytical Neurochemistry
Short description
In our group we develop and advance molecular imaging techniques based on both mass spectrometry, functional microscopy and chemical nanoscopy for probing chemical plaque pathology in brain tissue and cell models. The major goal is to use these techniques for delineating molecular mechanisms of abnormal protein oligomerization and aggregation underlying Alzheimer’s disease pathology.
Overview
The pathological mechanisms underlying Alzheimer’s disease (AD) remain largely unresolved. A seminal pathological feature in AD and other neurodegenerative disorders is the progressive accumulation and aggregation of misfolded proteins into intra and extracellular deposits.
The research in our group comprises both the development of innovative chemical imaging techniques including imaging mass spectrometry and chemical nanoscopy, with the aim to elucidate molecular aspects of amyloidosis associated with neurodegenerative processes particularly in Alzheimer’s disease.
More specifically, our research focuses on delineating beta-amyloid (Aβ) peptide aggregation dynamics, plaque associated lipids, and proteins in AD.
The primary aim is to understand the mechanism behind transforming beta-amyloid (Aβ) from being a non-toxic monomer to a neurotoxic oligomeric species that ultimately leads to nerve cell death and cognitive decline.
In this research we e.g., aim to identify chemical factors underlying Aβ plaque polymorphisms. Aβ deposits with diffuse morphology occur e.g., also in individuals without dementia (cognitively unaffected, amyloid positive CU-AP). While mature plaques are solely observed in AD. This suggests that formation of mature plaques is critical to developing AD. It has been suggested that diffuse plaques represent a preceding state of mature plaques. It is therefore of particular interest of our research to characterize the chemical phenotype (peptide truncation, associated lipids) of different plaque phenotypes in CU-AP in AD and to further elucidate how these plaque characteristics correlate with spread of AD pathology and cognitive decline.
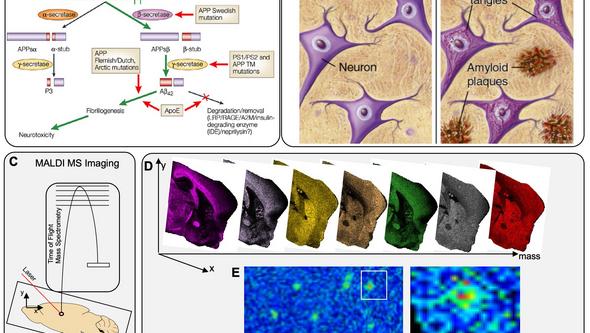
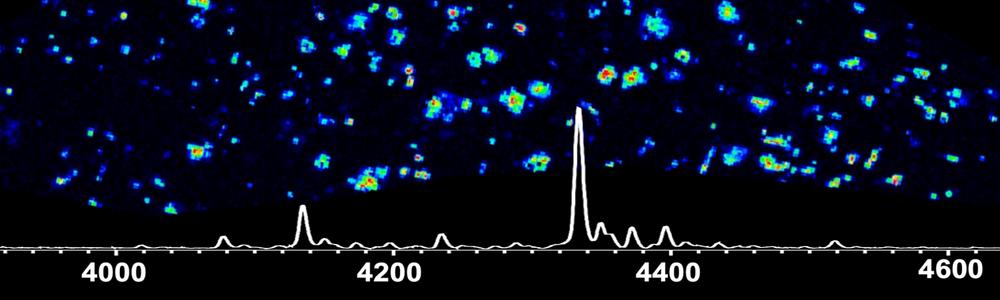
Figure above, enlarged. Aβ pathology and Mass Spectrometry Imaging (MSI). (A) Overview of Amyloid pathway of APP and influence of genetic factors leading to secretion of Aβ42 and aggregation. (B) Pathological hallmarks of AD in the brain: amyloid plaques and neurofibrillary Tau tangles. (C) MALDI MSI uses a laser for ionization of intact Aβ peptides that can be imaged across brain sections at 10μm. (D) Single ion images can be generated for different peptides in parallel, including and Aβ peptide outlining spatial peptide localization across different brain regions and within single Aβ plaques.
1. Multimodal Imaging of Amyloid Plaque Pathology
Over the last years, our group has been working on different molecular imaging modalities to probe chemical and structural aspects of amyloid pathology in AD. Here, we have used a large-scale effort in implementing multimodal chemical imaging technologies for comprehensive analysis of distinct plaque morphotypes in brain tissue. We developed a novel imaging paradigm that combined structure specific fluorescent amyloid dyes with fluorescent imaging in combination with imaging mass spectrometry. Using this strategy, we were able to delineate the chemical traits, peptides and lipids, associated with plaque polymorphism in transgenic AD mice.
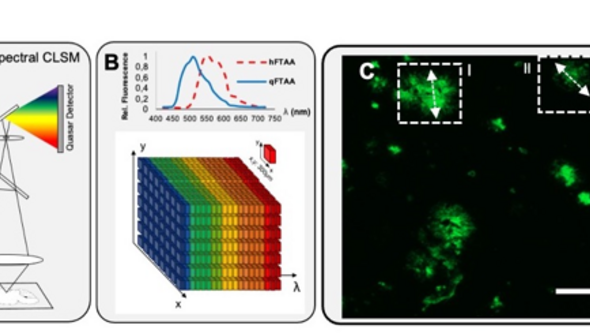
Characterization of Aβ polymorphism using functional amyloid imaging. (A) Plaque classification using structure-sensitive LCO probes (q/h-FTAA with different emission properties) that can be delineated with (B) hyperspectral microscopy. (C,D) Linear unmixing allows structural annotation based on the LCO emission profiles. A characteristic blue shift at the core indicates q-FTAA-binding mature amyloid fibrils (D-I).
A. Michno W, Kaya I, Nyström S, Guerard L, Nilsson KPR, Hammarström P, Blennow K, Zetterberg H, Hanrieder J.* “Multimodal Chemical Imaging of Amyloid Plaque Polymorphism Reveals Aβ Aggregation Dependent Anionic Lipid Accumulations and Metabolism” 2018 Anal Chem. 90(13):8130-8138. PMID: 29856605
B. Michno W., Wehrli P., Zetterberg H., Blennow, K., Hanrieder J.* “GM1 locates to mature amyloid structures implicating a prominent role for glycolipid-protein interactions in Alzheimer pathology” 2018 Biochim. Biophys. Acta 2019 (1867(5):458-467. PMID:30273679
2. Understand Amyloid Polymorphism in Human Brain
We have integrated these hyperspectral chemical imaging tools to investigate plaque polymorphisms in postmortem brain tissue. Here, we identified the distinct Aβ species associated with amyloid polymorphism in brain tissue from individuals with sporadic AD (s-AD) and CU-AP. We found that maturation of diffuse into cored plaques correlated with increased Aβ1-40 deposition, where Aβ1-40 was found to aggregate at the core structure of mature plaques, whereas Aβ1-42 localizes to diffuse amyloid aggregates. Moreover, we observed that diffuse plaques have increased pyroglutamated Aβx-42 levels in s-AD but not CU-AP, suggesting an AD pathology–related, hydrophobic functionalization of diffuse plaques facilitating Aβ1-40 deposition. These results suggest that diffuse deposits are immature precursors of cored plaques and that pyroglutamation of N-terminal Aβx-42, and Aβ1-40 deposition, are potentially critical events in priming and maturation of pathogenic Aβ from diffuse into cored plaques. These processes could underlie development of neurotoxic plaque pathology in AD and could hence provide a mechanistic target for potential intervention.
A. Michno W, Nyström S, Lashley T., Wehrli P., Brinkmalm G., Kaya I, Brinet, D., Guerard L, Syvänen S., Sehlin D., Nilsson KPR, Hammarström P, Blennow K, Zetterberg H, Hanrieder J.* “Pyroglutamation of Aβx-42 followed by Aβ1-40 deposition underlie priming and maturation of diffuse into cored plaque morphotypes in progressing Alzheimer’s disease pathology” 2019 J Biol Chem 294(17):6719-6732. PMC6497931
B. Pagnon de la Vega M., Giedraitis V., Michno W. Kilander L, Guener G, Zielinski M, Brundin RM, Danfors T, Söderberg L, Alafuzoff I, Nilsson LNG, Erlandsson A, Willbold D, Schröder GF, Müller SA, Hanrieder J, Lichtenthaler SF, Lannfelt L, Sehlin D and Ingelsson M. “The Uppsala APP mutation causes early onset Alzheimer’s by altering APP processing and increasing amyloid-β fibril formation” Science Transl Med 2021 13(606):eabc6184. PMID:34380771
3. Following Amyloid Aggregation Dynamics In Vivo
Our lab has pioneered the use of multimodal imaging experiments aim to characterize chemical and structural features of plaque pathology relevant in AD pathogenesis. Amyloidogenic protein aggregation, however, has to be considered as a highly dynamic process, which requires experimental setups that allow to monitor these processes as a function of time as the fibrillization dynamics of Aβ are of integral relevance in AD pathogenesis.
To address this issue, our group has developed a multimodal chemical imaging paradigm: imaging mass spectrometry towards stable isotope labelling to measure protein aggregation kinetics (iSILK), which we use to quantify amyloid peptide aggregation dynamics in genetic mouse models of AD. We have recently completed and published a first iSILK study where we follow plaque formation in APP KI mice.
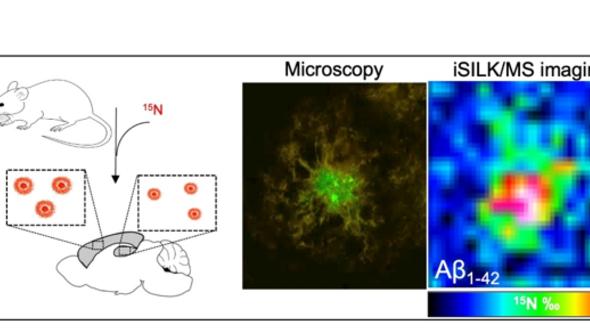
Principle of iSILK. (A) Mice are metabolically labeled with 15N diet leading to incorporation into Aβ plaques that are delineated with MSI (C) and correlative, hyperspectral microscopy (B).
These iSILK experiments allowed for the first time to visualize aggregation dynamics of different Aβ peptides within single plaques and across different brain regions in evolving plaque pathology from early deposition to later plaque growth. Specifically, we have demonstrated, using 6-week-old APPNL-G-F mice, that the method of feeding the stable isotopes works and leads to detectable levels of label within the plaques for meaningful analysis. To establish the early events of Aβ aggregation and heterogenous plaque formation, we performed a series of elaborate, complementary PULSE/CHASE setups. We found that Aβ pathology in APPNL-G-F mice precipitates in the cortex by forming small dense-core deposits (5µm in size) consisting of Aβ 1-42. The results show that plaques in APPNL-G-F mice form via early deposition of Aβ1-42 into compact core which is followed by plaque growth by homogenous deposition throughout the plaque. Later events in early plaque pathology involve deposition in the hippocampus, and secretion and deposition of Aβ1-38.
A. Michno, W., Stringer, K. S., Enzlein, T., Passarelli, M. K., Escrig S., Blennow, K., Zetterberg, H., Meibom, A., Hopf, C., Edwards, F. A., Hanrieder, J. “Following spatial Aβ plaque aggregation dynamics in evolving Alzheimer’s pathology by imaging stable isotope labelling kinetics (iSILK)” Science Advances 2021 7(25):eabg4855. PMC8208724
B. Michno W, Wehrli P, Sehlin D, Syvänen S, Zetterberg H, Blennow K, Hanrieder J. “Chemical imaging of evolving amyloid plaque pathology and associated Aβ peptide aggregation in transgenic AD Mice” J.Neurochem 2020 152(5):602-616. PMID: 31605538
Group members

Principal Investigator
Group members
- Maciej Dulewicz, Post Doctor
- Durga Jha, Post Doctor
- Sophia Weiner, Post Doctor
- Junyue Ge, Doctoral student
- Jack Wood, Doctoral student (University College London)
- Sneha Desai, Doctoral student (University College London)
- Aleksandra Antic, Doctoral student
- Alicja Szadziewska, Doctoral student
- Sofia Johansson, Doctoral student
- Lydia Fenson, Doctoral student
Organisational unit
Psychiatry and Neurochemistry
Institute of Neuroscience and Physiology


