SATMEG
Sahlgrenska Translational Melanoma Group (SATMEG) is a collaboration between researchers who hope to tailor individually designed therapies for patients suffering of melanoma in Western Sweden.
About Melanoma
Malignant melanoma is a cancer that arises from the pigment cells, the melanocytes. The most common site for the disease is the skin, where it can develop spontaneously or from a preexisting lesion such as a congenital, acquired, or atypical (dysplastic) nevus. Melanocytes also exist in other places in the body and melanoma can therefore also develop in the mucosal epithelium, retinas, and leptomeninges. The most common site is the skin and melanoma is the deadliest form of skin cancer.
Melanoma is potentially curable with surgical excision of early, thin lesions. Therefore, prompt detection, diagnosis, and adequate removal of such lesions are of utmost importance. If the disease progresses undetected it has a very poor prognosis and stage IV (distantly metastatic) disease has a 10-year survival of only 7-8 %.
Genetics of melanoma
A great deal of research has gone into identifying the genetic cause of melanoma. Mutations in growth-factor signaling are seen in most melanoma. These include NRAS mutations (20-25 %; codon 12, 13 and 61) and BRAF (50-65 %; V600E/K) for skin melanoma, but KIT mutations in mucosal and non-sunexposed skin melanoma (acral melanoma; 10-20 %). In eye melanoma, a rare disease, mutations in GNAQ and GNA11, which encode G-proteins are seen in a mutually exclusive manner.

Melanoma frequently also carry mutations in tumor suppressor genes. The most mutated tumor suppressor in other human cancers, TP53, is only mutated in about 5-10 % in melanoma. CDKN2A, encoding p16 and p14ARF, is mutated in many cases and also in some families with a predisposition to melanoma. RB, in the same pathway, is mutated in rare instances.
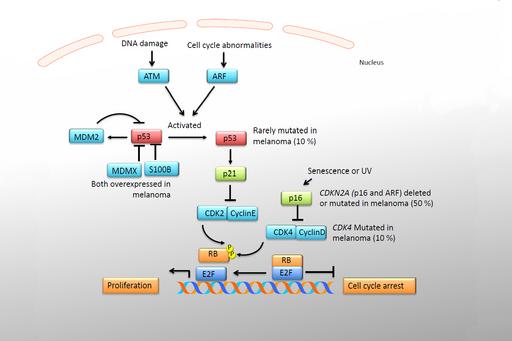
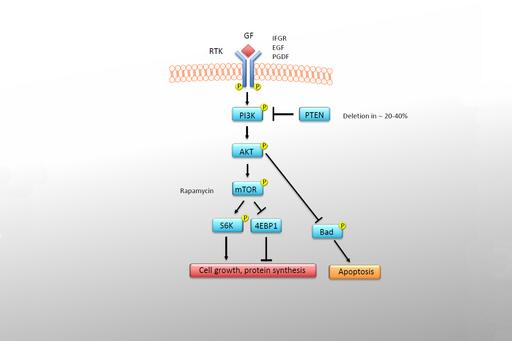
PTEN is mutated in 10 % of cases but low protein expression has also been observed and is associated with poor prognosis. In eye melanoma, BAP1 is a mutated in both familial as well as somatic cases.
Treatment
Historically melanoma has been regarded as a very difficult to treat disease and decades of clinical trials have failed to significantly improve overall survival of metastatic stage IV skin melanoma. Recently, targeted therapies and immune therapies have changed this but much work still remains until melanoma can be regarded as a controllable disease.
In the case of BRAF-mutant tumors, impressive therapeutic responses have been achievable by the development of mutation-specific inhibitors. Vemurafenib and Dabrafenib were the first FDA/LV-approved therapies targeting the gene product of the mutated BRAF allele. The drugs improve both overall survival (OS) and progression-free survival (PFS) but relapse due to resistance eventually develops. The reasons for the developed resistance are many. Since most mutations affect the same pathway it is noteworthy that combination treatment of B-Rafand MEK inhibitors has shown promising results in clinical trials.
It is known for a long time that melanomas are highly immunogenic because of the presence of tumor antigens, melanocyte antigens and aberrantly expressed or mutated antigens. Because of this, immune therapies have been developed with the aim of boosting the immune system reaction against the melanoma.
Current therapies include IFN and IL-2, the FDA-approved ipilimumab and anti-PD1 (in clinical trials). Ipilimumab is a monoclonal antibody directed against CTL-A4 on cytotoxic T-cells. Binding of ipilimumab blocks dampening of cytotoxic T-cell activity and results in enhanced anti-tumoral activity in patients alone or in combination with the alkylating agent dacarbazine.
A similar mechanism involving differentsurface molecules is operational with anti-PD1 or anti-PD1L therapies. Combinations of these therapies are tested in clinical trials and hope to achieve higher response rates than the individual therapies.
SATMEG - Purpose and aims
The discovery of recurrent activating mutations in BRAF, the development of targeted therapies against the mutated B-Raf protein and observing impressive clinical response rates with Vemurafenib and Dabrafenib have revolutionized melanoma treatment and brought huge attention to the disease. For all the right reasons, most current research is focused on the MAPK-pathway and different means to block resistance development and achieve durable responses to B-Raf inhibitors.
Knowing that this pathway eventually ends up in activating transcription factors like c-Myc, SATMEG would like to translate the knowledge gained about pathways operating downstream and parallel to that transcription factor. By utilizing genetically-engineered and melanoma patient-derived xenograft mouse models we hope to tailor individually designed therapies for patients suffering of melanoma in Western Sweden.
SATMEG has already initiated two clinical trials for metastatic UVM patients: SCANDIUM (clinical PI: co-applicant Roger Olofsson Bagge) and PEMDAC (clinical PI: co-applicant Lars Ny).
SCANDIUM trial
In this trial, isolated liver perfusion, a surgical oncology method is compared to best-alternative-care in a phase III academic trial. During the surgical procedure, biopsies are given to the lab so that we can analyze the tumors’ genetics and generate PDX models. Since biopsies are generally not taken from UVM patients’ liver metastases, this is a truly unique possibility.
PEMDAC trial
In this trial, epigenetic therapy (HDAC inhibitor entinostat) is combined with immune therapy (PD1 antibody pembroluzimab). The rationale for this trial comes from pre-clinical data from the lab showing that it is possible to enhance the expression of antigen-presenting molecule HLA of uveal melanoma by HDAC inhibitors. There are also likely other mechanisms that could enhance immunity and translational efforts of this trial will elucidate mechanism of action if responses are seen.
Core members
Jonas Nilsson, PhD
Professor in Experimental Cancer Surgery
Roger Olofsson Bagge, MD/PhD
Professor & Team leader Breast and Melanoma Surgery
Lars Ny, MD, PhD
Associate professor & Department Head in melanoma oncology
Ulrika Stierner, MD, PhD
Consultant oncologist
Lisa Nilsson, PhD
Senior staff scientist
Collaborators
Local
Erik Larsson Lekholm, PhD, Associate professor
Peter Naredi, MD/PhD, professor
Jan Lötvall, MD/PhD, professor
Ulf Yrlid, PhD, Associate professor
Jerker Mårtensson, PhD, Associate professor
Ola Nilsson, MD/PhD, professor
National
Thomas Helleday, KI
Nelson Gekara, Umeå University
Martin Bergö, KI
International
Ulrich Keller, TU Munich, Germany
Marco Donia and IngeMarie Svane, Herlev Hospital Denmark
Piro Lito, MSKCC, New York