TAT - Chemistry
Short description
The standard production of 211At is by irradiating stable bismuth with alpha-particles produced in a cyclotron, and date back to the discovery of the nuclide in 1940 by Corson et al (1). Since 1994 different cyclotron operators have provided the TAT group with 211At utilizing the production method mentioned above.
Today we receive the irradiated target on a regular basis from the PET and Cyclotron Unit at Copenhagen University Hospital in Denmark. From the target we recover astatine-211 through dry-distillation and convert the nuclide into a chemically useful form. Since 2014 this procedure is done using an automatic equipment developed in-house. As of 2018, targets for irradiation are also procued in-house by the TAT group and send off to Denmark for irradiation.
The astatine can now be used to label different carrier vectors such as antibodies, antibody fragments, peptides or small molecules. The labeling chemistry is most often based on organic tin derivatives, a method first developed by Zalutsky and co-workers (2). During the years we have further developed the tin-based labeling chemistry by changing the reaction route to improve radiochemical yields (3) investigating the stability of the prefabricated immunoconjugates used in this route (4) and also developing a new tin-based reagent for site selective labeling (5), Figure 1.
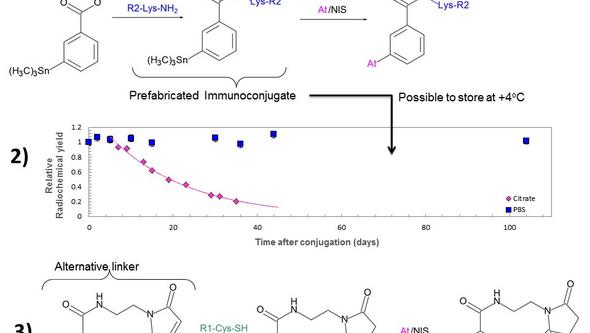
In addition to astatine we have access to another interesting alpha particle emitting radionuclide, Bismuth-213, through a collaboration with the Institute of Transuranic Elements (ITU) in Karlsruhe, Germany. They provide us with 225Ac/213Bi generators from which we can elute 213Bi in a pure chemical form for subsequent labeling to antibodies. The labeling is performed using commercially available bifunctional chelates.
The half-life of 211At (7.2 h) and especially 213Bi (46 min) is generally too short for conventional radioimmunotherapy, except for a few special applications such as blood-born or intracavitary cancer treatments. In order to circumvent the unfavourable pharmacokinetics of radiolabeled antibodies, various so called pretargeting techniques have been suggested (8-10).
A successful pretargeting protocol will improve the tumor-to-normal tissue absorbed dose ratio for all types of applications involving antibodies for tumor targeting together with radionuclides with short half-lives. We are currently working on several different projects on pretargeting such as Poly-L-lysine based scaffolds functionalized with (strept)avidin/biotin (Figure 2) or TCO/tetrazine as well as bifunctional astato triazol molecules and imminobiotin based systems.
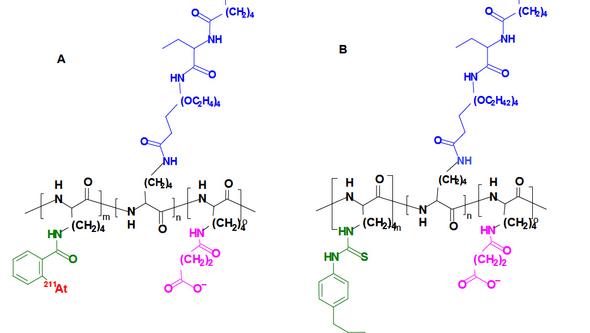
succinic acid residue following charge modification.
- D.R. Corson, K.R. MacKenzie, Phys. Rev. 58. 672-678 (1940).
- M.R. Zalutsky, A.S. Narula, Appl. Radiat. Isot. 55. 157-160 (2001).
- S. Lindegren, S. Frost, T. Bäck, E. Haglund, J. Elgqvist, H. Jensen, J. Nucl. Med.49. 1537-1545 (2008).
- E. Aneheim, J. Halleröd, S. Holgersson, P. Albertsson, S. Lindegren. Canc. Biother. Radiopharm. 41-45 (2015)
- E. Aneheim, A. Gustafsson, P. Albertsson, T. Bäck, H. Jensen, S. Palm, S. Svedhem, S. Lindegren. Bioconj. Chem. 688–697 (2016)
Related Links
- American Association for Clinical Chemistry
- American Chemical Society (External link)
- Chemistry Dictionary (External link)
- ChemistryGuide.org (External link)
- ChemPlace.com (External link)
- European Association of Nuclear Medicine (External link)
- Nature - Chemistry (External link)
- NIST Chemistry WebBook (External link)
- Nobel Prizes in Chemistry (External link)
- Periodic Table (External link)
- Radiochemistry Society (External link)
- Royal Society of Chemistry (External link)
- Science Daily - Chemistry (External link)
- Society of Nuclear Medicine (External link)
- Swedish Chemical Society (External link)